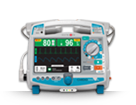
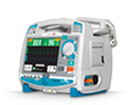
Cardioverter/Biphasic Defibrillator
What all emergency equipment should be: complete, advanced, reliable and easy to use.
Intuitive, easy to use commands, based on the standard three operating steps.

Large variety of optional items, allowing assembly according to your specific needs or future updating.

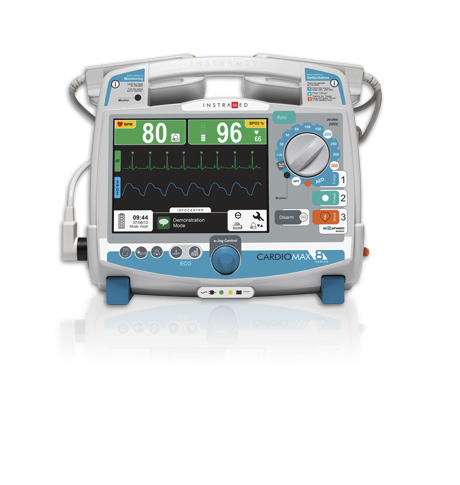
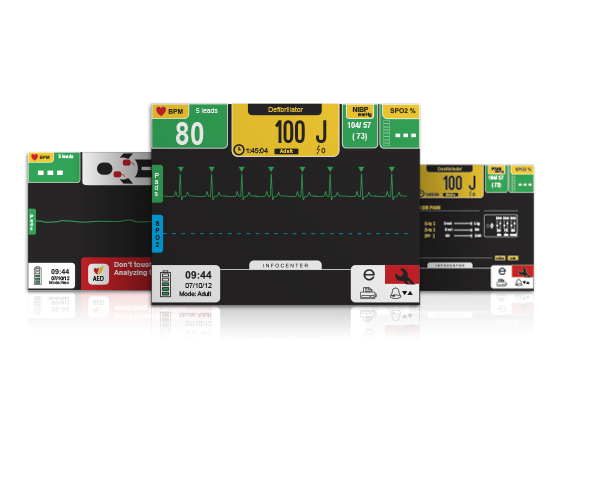
Advanced, practical user interface: shows vital information clearly and provides easy access to functions and settings.
Integrated adult/child paddles for monitoring and two phase shock of up to 360 joules.

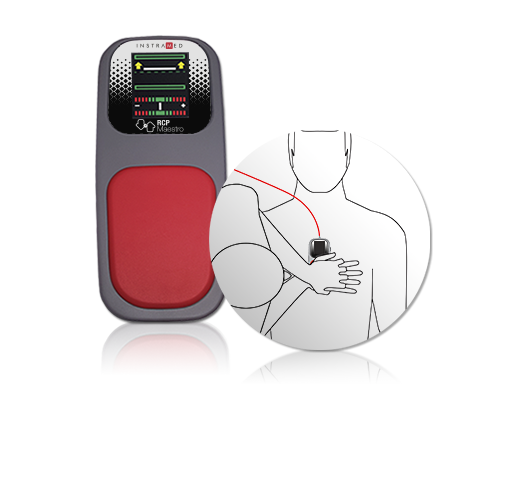
CPR Maestro: accessory for assistance and feedback of cardiopulmonary resuscitation.
DESCRIPTION
PRACTICAL
- Light.
- Strong, comfortable strap.
- Ready to use in less than 6 seconds.
- Designed without sharp edges, ideal for emergency transport.
- Biphase power delivery of up to 360 Joules.
- The battery, which is easy to replace, allows more than 100 shocks.
SMART
- Interface that automatically adjusts to the number of parameters, presenting the important information in a clearer and more organized way.
- Smart monitoring alarms.
EASY TO USE
- All operations are concentrated in only two buttons.
- Easy operation - 1, 2, 3 standard.
- Integrated adult/children pads.
- Quick access to main functions.
VERSATILE
The CardioMax can become even more complete and convenient through various optional items available that help accompany high-risk patients:
- AED Module - Automated External Defibrillator
- Real Time Check technology
- Sudden Death Prevention technology
- Charge Auto-Sequencing technology
CHARACTERISTICS
- Real Time Check Technology (RTC)**
- Cardiopulmonary Resuscitation Accessory(CPR Maestro)**
- Automated External Defibrillator Mode (AED)
- Sudden Death Prevention Mode (SDP)
- Electrocardiogram (ECG) up to 12 derivations
- Oximetry (SpO2)
- Non invasive pacemaker
- Non-invasive pressure (NIBP)
- Capnography (EtCO2)
- Printer
- Removable rechargeable battery
*Some items are optional.
**Pre-release product. Please verify availability.
ADVANCED TECHNOLOGIES
RTC - Real Time Check
Sudden Death Prevention
Charge Auto-Sequencing
Reliability is the most important aspect for equipment designed to save lives. With the new RTC (Real Time Check) technology, CardioMax performs constant self-diagnosis and reports, in advance, if there is any maintenance action to be done, guaranteeing that it will always be available for immediate use.
The Sudden Death Prevention (SDP) technology allows CardioMax to monitor the patient continually and identify the beginning of a Ventricular Fibrillation or Rapid Ventricular Tachycardia episode. In this situation, the equipment activates a visual and sound alarm, allowing the patient to be treated with shock in a much shorter time, significantly increasing the chances of reversing cardiorespiratory arrest.
New Charge Auto-Sequencing function - When enabled, applies charges pre-configured by the user for the first, second and third shocks without the need to change the selector manually.
CPR MAESTRO
ASSISTANCE AND GUIDANCE FOR CPR
CPR Maestro is an accessory that helps and guides the application of CPR (Cardiopulmonary Resuscitation), making the process much more efficient and reliable, considerably increasing the chances of cardiopulmonary arrest reversal.


RELIABLE, EFFICIENT AND ERGONOMIC
Positioned on the patient's chest, the CPR Maestro provides real-time feedback, guiding the rescuer by voice and visual indications on the device's integrated LCD display.
Its sophisticated sensor system reliably and accurately indicates the optimal frequency, strength and interval for the procedure, resulting in increased patient survival.
Its ergonomic design and the use of flexible materials in contact areas facilitate the use and visualization of information, minimizing the effects of treatment on the patient.
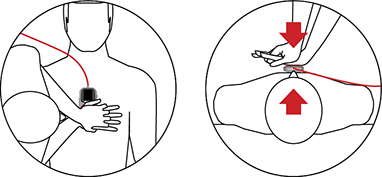
THE IDEAL PARTNER FOR YOUR CARDIOMAX
Developed for use in conjunction with the Instramed defibrillators line, the CPR Maestro features a simple interface that visually guides the rescuer. On compatible Instramed devices, this information and guidance are shown expanded on the defibrillator screen, providing user convenience and safety. After use, CPR Maestro also offers usage statistics for further evaluation of the procedure.

TECHNICAL INFORMATION
GENERAL SPECIFICATIONS
- Dimensions with pads:
- 30,0 cm (11.81 in) length.
- 21,5 cm (8.46 in) depth.
- 28,0 cm (11.02 in) height.
- Weight:
- Device - 5.15 kg (11.35 lbs).
- NiMH battery - 1.10 kg (2.43 lbs).
- Li-Ion battery - 0.60 kg (1.32 lbs).
- External pads - 0.85 kg (1.87 lbs).
- Complete set (NiMH battery) - 7.10 kg (15.66 lbs) (except NIBP).
- Complete set (Li-Ion battery) - 6.60 kg (15.66 lbs) (except NIBP).
- Electrical:
- AC: 100 to 220 VAC, 50/60 Hz (automatic selection).
- DC external: 11 to 16 VDC.
- Removable rechargeable battery:
- Type: Li-Ion, 14.8 VDC 4.4 A/h.
- Duration: Battery with full charge - 3 hours in monitor mode, without printer, or a minimum of 140 shocks at 360 Joules or a minimum of 200 shocks at 200 joules.
- Battery full-charge time (when fully unloaded): 4h 30min.
- OPTIONAL BATTERY**:
- Type: NiMH, 14.4 VDC 4.5 A/h.
- Duration: Battery with full charge - 3 hours in monitor mode, without printer, or a minimum of 140 shocks at 360 joules or a minimum of 200 shocks at 200 joules.
- Battery full-charge time (when fully unloaded): 8 hours.
- **Consult availability.
- Memory:
- Type: Nand Flash.
- Capacity: 2 Mbytes.
- Patients stored >150 patients. - Storage: 15 seconds of ECG when in shock, physiological alarm and panel events.
- ECG: 2 continuous hours of ECG curve recording, when in AED mode.
- RTC – Real Time Check (available when equipped with Li-lon optional battery):
- Defibrillation self-test, battery level, connected pads, power source connection check. Check is performed 3 times which are set in advance. This information is wirelessly transmitted to a PC with RTC System software installed and within range of the network.
ENVIRONMENTAL SPECIFICATIONS
- Temperature:
- Operational: 0 to 50°C.
- Storage: -20 to 50°C.
- Humidity:
- Operational: 10 to 95% RH, without condensation.
- Storage: 10 to 95% RH, without condensation.
- IP Rating: IPX1.
DEFIBRILLATOR
- Waveform:
- Biphasic truncated exponential. Waveform parameters adjusted in terms of patient’s impedance.
- Shock application:
- By means of multifunctional pads (adhesive) or defibrillation pads.
- Adult/external defibrillation:
- Scales:1,2,3,4,5,6,7,8,9, 10, 20, 30, 50, 80, 100, 150, 200, 250, 300 and 360 Joules (maximum power can be limited to 200 J). Maximum power limited to 50 J with internal or children’s pads.
- Controls: On/Off button, charge, shock, synchronism.
- Power selection: Therapy button on the front panel.
- Charge control: Button on the front panel, button on the external pads.
- Shock control: Button on the front panel, buttons on the external pads.
- Synchronized control: SYNC button on the front panel.
- Charge Auto-Sequencing:
- When enabled, it charges power previously set by the user for the first, second and third shocks, with no need to manually adjust the selector.
- Charge indicators:
- Sound signal of equipment being charged.
- Sound signal of completed charge.
- LED on external pads and charge level indicated on the display.
- Maximum charging time:
- (200J): mains supply and battery < 4s.
- (360J): mains supply and battery < 6s.
- Electrode size:
- Adult: 10.3 cm (4.05 in) x 8.5 cm (3.34 in). Contact area: 81.9 cm2 (12.69 in2)
- Children: 4.5 cm (1.77 in) x 4.0 2 cm (1.57 in). Contact area: 18 cm2 (2.79 in2)
- Cardioversion:
- < 60 ms.
- Pads (options):
- Adult and child external (included).
- Adult and child internal (optional).
- Multifunctional for pacemaker, monitoring and defibrillation (optional).
- Multifunctional extension (optional).
AED MODULE
- Functional characteristics:
- Voice instructions, visual indications, CPR instructions, USB 2.0. Multilanguage, Sudden Death Prevention Technology (SDP).
- USB:
- USB 2.0 for transfer of the electrocardiogram stored in AED mode to a compatible PC.
- SoftDEA:
- Software for viewing the data transferred to the PC.
EXTERNAL PACEMAKER (OPTIONAL)
- Modes:
- Demand or fixed.
- Amplitude:
- From 5 mA to 200 mA (resolution of 5 mA), accuracy 10%.
- Pulse width:
- 20 ms (± 1 ms).
- Frequency:
- From 30 ppm to 180 ppm (increments of 5 ppm), accuracy ± 2%.
- Refractory period:
- 340 ms (from 30 to 80 ppm).
- 240 ms (from 90 to 180 ppm).
NIBP (OPTIONAL)
- Operating principle:
- Oscillometric.
- Automatic measurement mode:
- 1, 2, 3, 4, 5, 10, 15, 30, 60 and 90 minutes.
- Manual:
- One measurement.
- Measurement interval:
- Adult:
- Systolic: 40 - 260 mmHg.
- MAP: 26 - 220 mmHg.
- Diastolic: 20 - 200 mmHg.
- Pediatric:
- Systolic: 40 - 160 mmHg.
- MAP: 26 - 133 mmHg.
- Diastolic: 20 - 120 mmHg.
- Neonate:
- Systolic: 40 - 130 mmHg.
- MAP: 26 - 110 mmHg.
- Diastolic: 20 - 100 mmHg.
- Overpressure protection by hardware:
- Adult: 300 ± mmHg.
- Neonate: 150 ± mmHg.
- Overpressure protection by hardware:
- Adult: 300 ± mmHg.
- Neonate: 150 ± mmHg.
- Resolution:
- 1 mmHg.
CPR Maestro**
- Accessory for Cardiopulmonary Resuscitation
- Feedback of the thoracic compressions.
DISPLAY
- Battery level indicator:
- Yes.
- Size:
- 128.2 mm x 170.9 mm.
- Diagonal:
- 8.4”.
- Type:
- Color LCD TFT.
- Resolution:
- 640 x 480 pixels (VGA).
- Scan speed:
12.5; 25 and 50 mm/s.
ECG (supports up to 12 simultaneous derivations when equipped with optional cable)
- Inputs:
- 3 or 5 lead ECG cable.
- 10 lead ECG cable (optional).
- External pads.
- Multifunctional pads.
- Range:
- 15 to 350 BPM.
- Precision:
- ± 1 BPM from 15 to 350 BPM.
- Rejection in common mode:
- Greater than 90 dB, measured according to AAMI standards for heart monitors (EC 13).
- Sensitivity:
- 5, 10, 15, 20, 30 and 40 mm/mV.
- AC line filter:
- 60 Hz or 50 Hz.
- ECG response frequency:
- Diagnostic mode - (0.05 -100 Hz).
- Monitor Mode - (1-40 Hz).
- Patient insulation:
- Defibrillation proof.
- ECG: CF Type.
- SpO2: CF Type.
- Loose electrode:
- Identified and shown with low level alarm.
- Time to restore ECG baseline after defibrillation:
- ≤ 3 seconds.
SpO2 (OPTIONAL)
- SpO2 range:
- 0 to 100%.
- Pulse range:
- 30 to 250 BPM.
- SpO2 precision:
- ± 2 % from 70 to 100%.
- ± 3 % from 50 to 69%.
- Pulse precision:
- ± 2 BPM.
- Scan speed:
- 12.5; 25 e 50 mm/s.
CAPNOGRAPHY (OPTIONAL)
- Measurement range CO2:
- 0 – 99 mmHg.
- Precision:
- ± 2 mmHg (0 – 38 mmHg).
- ± 5% + 0.08% for each 1mmHg above 38 mmHg (39 – 99mmHg).
- Consumption:
- 1.5 W.
- Compensation:
- BTPS, N2O, O2.
PRINTER (OPTIONAL)
- Prints up to three simultaneous derivations.
- Type:
- Thermal.
- Weight:
- 0.4 kg.
- Speed:
- 12.5; 25 or 50 mm/s with precision of ± 5%.
- Paper size:
- 58 mm (width) x 15 m (maximum length).
STANDARDS
- NBR IEC 60601-1
- NBR IEC 60601-1-2
- NBR IEC 60601-1-6
- NBR IEC 60601-1-8
- NBR IEC 60601-2-4
- NBR IEC 60601-2-27
- NBR IEC 60601-2-30
- NBR IEC 60601-2-49
or equivalent IECs
Site language > Português |Español
Home | Company | Certifications
Products > CardioMax | I.on/I.on PRO | DualMax
Downloads > Brochures | Manuals | Softwares
Contact > Headquarters | International Distributors | Support
Copyright © - Instramed Indústria Médico Hospitalar Ltda. All rights reserved.